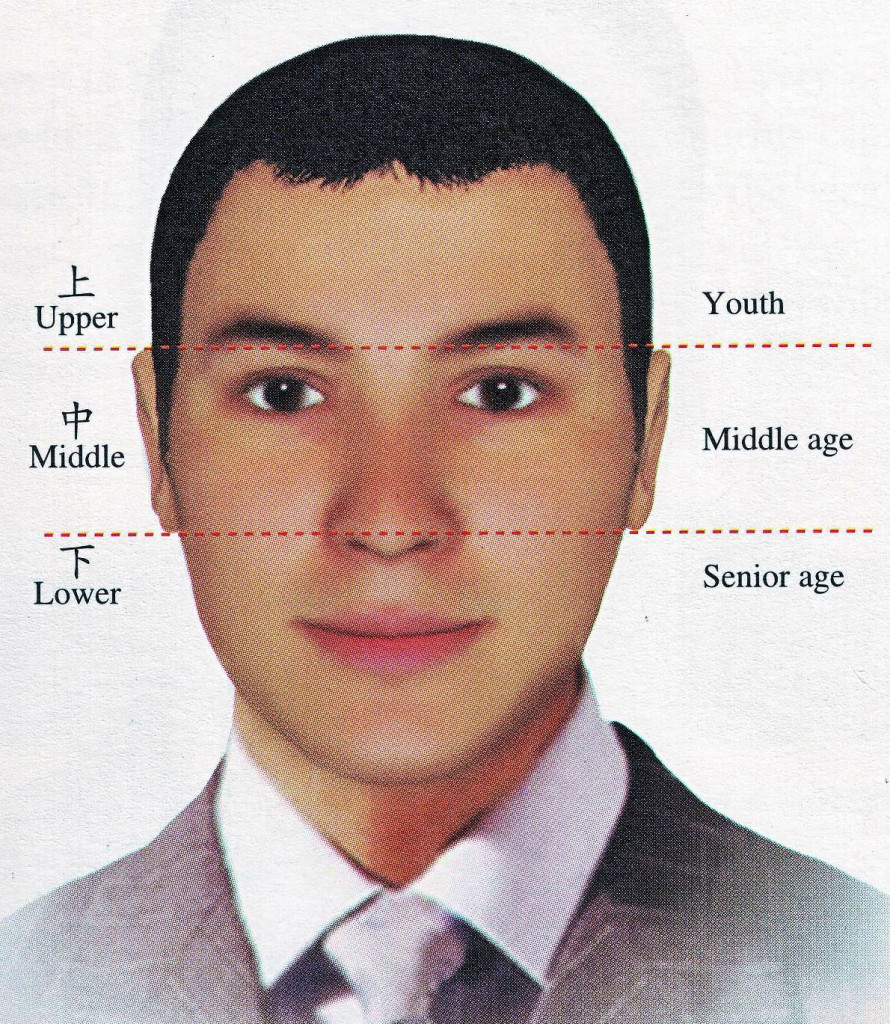
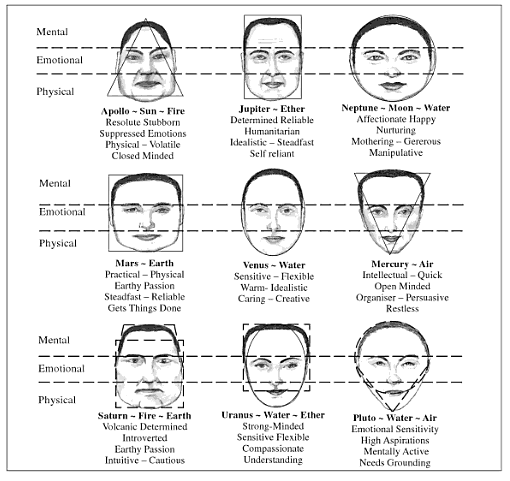
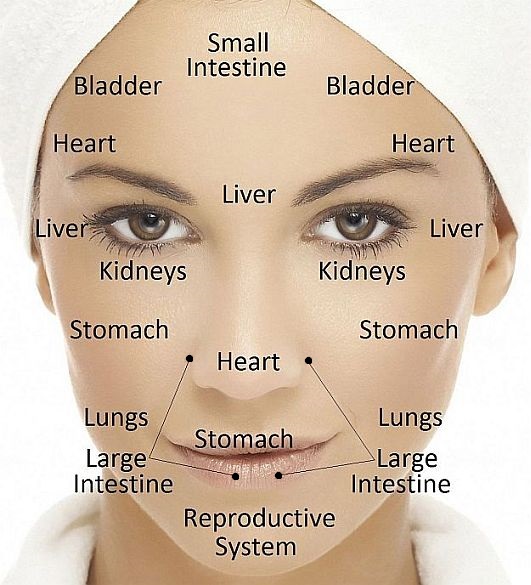
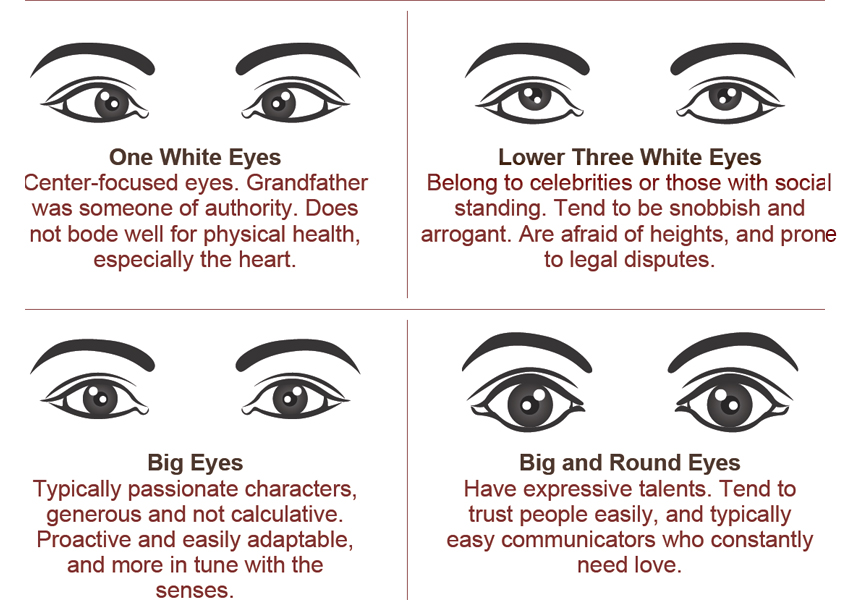
Ayurveda and Fall part 2
The Vata Dosha is the one connected with Fall lets find out what Vata is about:

In week 352 we are sharing therVata dosha predominates, movement and change are characteristic of the nature of Vata. You tend to always be on the go, with an energetic and creative mind. As long as Vata is in balance, you will be lively and enthusiastic, with a lean body, Energy that controls bodily functions associated with motion, including blood circulation, breathing, blinking, and your heartbeat.
• In balance: There are creativity and vitality.
• Out of balance: Can produce fear and anxiety.
Vata Predominant Types: Creative; Quick to learn and grasp new knowledge, but also quick to forget, Slender; Tall and a fast-walker; Tendency toward cold hands and feet, discomfort in cold climates; Excitable, lively, fun personality; Changeable moods; Irregular daily routine; High energy in short bursts; tendency to tire easily and to overexert; Full of joy and enthusiasm when in balance; Responds to stress with fear, worry, and anxiety, especially when out of balance; Tendency to act on impulse; Often have racing, disjointed thoughts; Generally have dry skin and dry hair and don’t perspire much.

Physical Characteristics
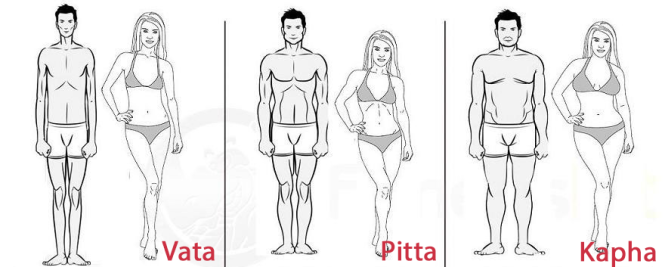
Those with a predominance of Vata dosha usually have a thin, light frame and excellent agility. Their energy comes in bursts and they are likely to experience sudden bouts of fatigue. Vata’s typically have dry skin and hair and cold hands and feet. They sleep lightly and their digestion can be sensitive. When the Vata dosha becomes imbalanced, it manifests in the body as weight loss, constipation, hypertension, arthritis, weakness, restlessness, and digestive challenges.
Emotional Characteristics
Vatas love excitement and new experiences. They are quick to anger but also to forgive. When Vata is in balance, they are energetic, creative, and flexible. They also take initiative and are lively conversationalists. When unbalanced, they are prone to worry and anxiousness and often suffer from insomnia. When they feel overwhelmed or stressed, their response is, “What did I do wrong?”
The influence of Vata’s ether and air contributions, we can feel light, carefree, and creative or spacey, scattered, and unstable. The etheric nature of Vata creates a sense of space, in which you may feel free or lost if it is not balanced. The airy aspect of Vata can inspire productivity or promote anxiety. Ayurveda teaches that like increases like much like the law of attraction, isn’t it?. If you are dominantly Vata by nature or are consistently influenced by Vata, you are more likely to experience the negative effects of excess Vata during the Vata season.
Who determines what Dosha we are and where it comes from?

Here is a quick test to find out what Dosha you are:
Here is a great site to determine what dosha you are: Chopra Dosha Quiz
Prakriti is our basic constitution. This is determined at the moment of conception and relates to your genetically inherited physical and emotional qualities. Prakriti specifically relates to those qualities, characteristics, and tendencies that are the stable makeup of ourselves. For instance, while you may experience temporary changes, like gaining or losing ten pounds, feeling nervous or irritable, developing a cold or flu, to mention a few, in the natural course of life you will never gain or lose five inches on your height or experience a change of eye color unless is a major affection, but not in our normal state of health.
Prakriti is enlivened and described by three main doshas or forces: Vata, Pitta, and Kapha. These are loosely translated as Air, Fire, and Earth, respectively. Each of us has all three doshas in our constitution, in our unique proportions.
In Ayurveda, seven dosha-predominant Prakritis are described: Vata-predominant, Pitta-predominant, Kapha-predominant;
Three dual Prakritis,
We’re two doshas are equally, or nearly equally predominant: Vata-Pitta predominant, Pitta-Kapha predominant
And Vata-Kapha predominant
And one Prakriti that has all three doshas equally prominent: Vata-Pitta-Kapha predominant.
Note: Even a thoughtful test cannot take the place of an evaluation by a qualified Ayurvedic practitioner. The results of this test may give a good indication of the primary doshas in your Prakriti but the evaluation of your practitioner may be more accurate.
Vata governs movement in the body, the activities of the nervous system, and the process of elimination.
Qualities of Vata:

• Cold
• Light
• Dry
• Irregular
• Rough
• Moving
• Quick
• Changeable
As the external environment changes during the Vata season, your internal environment can experience the same type of changes; dry leaves, dry skin; crackly leaves, crackly joints; shorter days, shorter attention span; colder days, colder extremities, windy days, windy bowels. The qualities of Vata dosha are found in the disorders that are common at this time of year. By observing the processes of Mother Nature, you can better understand the processes of your body, mind, and spirit.
Applying the Ayurvedic principle that opposite actions create balance, you can maintain balance during the Vata season by emphasizing lifestyle and food choices that are grounding, stabilizing, warming, moisturizing, and softening. You can stay calm and connected in this whirlwind season with a consistent practice that includes nourishing and protective measures. Ayurveda promotes simple and regular routines as having a deeper effect on balancing Vata than an ‘as needed’ approach.
Offered here are Ayurvedic recommendations for enjoying the Vata season with stability and serenity:
Fall is a time of transition. It is evident everywhere around you. Many trees and shrubs are quietly undressing in preparation for the winter. There is a subtle browning of the earth. Temperatures, which, just a few weeks ago were raging with the intense heat of summer, are beginning to hint at the telltale crispness of autumn. And there is the wind: slowly gathering strength, carrying the tides of winter on its breath. The autumn harbors a certain emptiness that can leave us feeling exposed and a little raw, but it is also filled with possibility—a time when we, too, can strip down to a quiet essence of being and savor the simplicity. The fall brings with it a predominance of air element and prana (the vital breath, the subtle essence of life) is abundant in the atmosphere. Autumn is dry, rough, windy, erratic, cool, subtle, and clear. These are all qualities shared by Vata dosha, and because like increases like, autumn is considered a Vata season. This same principle illustrates why taking a few simple steps to balance Vata this fall can be tremendously beneficial.
Ayurveda considers a seasonal routine an important cornerstone of health, year-round. Balancing the nature of your local climate with lifestyle choices that offset the potential for seasonally-induced imbalances is one of the simplest ways that you can protect your well-being. But keep in mind that the seasons vary widely from one place to another, as do the qualities that they engender. “Vata season” is whatever time of year most embodies the attributes that characterize Vata dosha: dry, light, cold, rough, subtle, mobile, and clear (or empty). Autumn is the classic Vata season. However, depending on where you live, the dry and expansive qualities of Vata may be prevalent components of your environment as early as summer, and the autumn may be followed by a very drying, cold, isolating, and/or windy winter.
Beginning to observe your environment from this qualitative perspective empowers you to respond to both daily and seasonal fluctuations in your local climate. The truth is that many of us adopt seasonally appropriate habits already, without even being conscious of doing so. For instance, summer is a time when we often enjoy salads and watermelon in abundance, both perfect antidotes to the heat and intensity of the summer. Whereas by October and November, we’re often baking delicious pumpkin bread and dining on hearty, grounding soups—foods that naturally subdue the dry, light, and erratic nature of the fall. By making diet and lifestyle choices that counter the effects of each season, you can better maintain your internal sense of equilibrium throughout the year
If we consider the Ayurvedic principle that opposites balance, Vata season (which is cool, light, dry, windy, and unpredictable) will be less aggravating if you fill it with warmth, oiliness, deep nourishment, loving relationships, and a sense of stability, routine, and groundedness. In addition, you may find it helpful to familiarize yourself with signs and symptoms of a Vata imbalance so that you are better prepared to address those immediately if they do arise. The following recommendations are appropriate for most people, but if you know your constitution or your current state of balance, you can tailor your seasonal routine appropriately. Below the general recommendations that follow, you will find links to more dosha-specific considerations.
Your diet is a powerful way to soothe Vata this fall. Substantive, oily, nourishing foods that are high in protein, high in fat, brought to life with warming, stimulating spices, and served hot, will go a long way toward maintaining your internal reserves of moisture and keeping you grounded through the Vata season. You’ll also want to favor the sweet, sour, and salty tastes. In general, eat mushy, soft foods, and garnish them generously with ghee or oil. Breakfasts of cooked grains—like oatmeal, tapioca, cream of rice, and cream of wheat—are perfect at this time of year. Lunches and dinners that include steamed vegetables, hearty grains, soups, and stews are grounding and moisturizing. If you eat meat and eggs, this is one of the best times of year to enjoy them. Dairy products and most nuts and seeds are also beneficial. In general, you’ll want to reduce your consumption of raw vegetables, cold and frozen foods, as well as the bitter, pungent, and astringent tastes. It is best to minimize light, cooling, and drying foods like broccoli, cabbage, cauliflower, sprouts, leafy greens, white potatoes, beans, popcorn, crackers, millet, and dried fruit. If you do eat these foods, eat them in moderation and make sure that they are soaked, well-cooked, or served with ghee.
You may find that, during the course of the fall, you’ll naturally want to increase your intake of food, but be careful to follow the lead of your appetite and digestion. This is also a great time of year to do a mono-diet type of cleansing. Vata requires adequate nourishment so it is best to avoid fasting.
The following is a list of ideal Vata season foods:
Fruits to Favor
Apples (cooked)
Avocados
Bananas
Dates
Figs
Grapefruit
Grapes
Lemons
Limes
Mangoes
Oranges
Papayas
Prunes (soaked)
Raisins (soaked)
Tangerines
Vegetables to Favor
Beets
Carrots
Chilies
Garlic
Okra
Onions
Pumpkins
Squash, Winter
Sweet Potatoes
Grains to Favor
Amaranth
Basmati Rice
Brown Rice
Oats
Quinoa
Wheat
Legumes to Favor
Kidney Beans
Miso
Mung Beans
Tur Dal
Urad Dal
Nuts and Seeds to Favor
All nuts and seeds are supportive of Vata season
Dairy to Favor
Butter
Buttermilk
Cheese
Cream
Ghee
Kefir
Milk (not cold)
Sour Cream
Yogurt
Animal Products to Favor (If You Eat Them)
Beef
Buffalo
Chicken
Crab
Duck
Eggs
Fish
Lobster
Oysters
Shrimp
Turkey
Venison
Oils to Favor
Almond Oil
Ghee
Olive Oil
Peanut Oil
Safflower Oil
Sesame Oil
Sweeteners
Honey
Jaggary
Maple Syrup
Molasses
Rice Syrup
Sugar (Raw)
Spices to Favor (All Spices Are Good for Vata Season)
Allspice
Anise
Asafoetida (Hing)
Basil
Bay Leaf
Black Pepper
Cardamom
Cinnamon
Clove
Cumin
Dill
Garlic
Ginger
Mustard Seeds
Nutmeg
Oregano
Paprika
Parsley
Rosemary
Saffron
Turmeric
Vata Season Lifestyle Choices
One of the most effective ways to support Vata is by establishing a daily routine. Try to do the same things (wake up, exercise, eat meals, go to bed, etc.) at roughly the same time each day. Set the tone for your day by rising early, taking full advantage of the silence, stillness, and peace that are intrinsic to the early morning hours. Then, you can calm your nervous system, awaken your tissues, and ground your energy by massaging your skin with warm, organic Sesame Oil. Follow this practice with a warm, relaxing shower, leaving a coat of oil on the skin to absorb throughout the day. Steam baths and humidifiers can help to preserve internal moisture as well. Some gentle yoga and ten to fifteen minutes of meditation will further your sense of stability and wellness. If you enjoy a little fragrance, vetiver, geranium, and citrus essential oils are very appropriate this time of year. Dress in autumn colors when appropriate—reds, yellows, oranges, and whites—and wear enough clothes that you stay warm throughout the day. When you step out into the elements, cover your head and ears to protect them from the biting wind and cold. If possible, minimize your exposure to drafts, loud noise, aggressive music, fast driving, and excessive sexual activity. Try to be in bed by 10 p.m. so that you get plenty of rest before dawn.
Vata Season Exercise
The best times of day to exercise are in the early morning and evening hours (6–10 a.m. and 6–10 p.m.). Vata is very easily aggravated by fast, mobile activities, so consider slow, gentle, strengthening forms of exercise instead. Walking, hiking, swimming, biking, yoga, and tai chi are good choices, provided they are done at an appropriate level of intensity. Ideally, exercise at about fifty to seventy percent of your capacity, breathing through your nose the entire time. And remember to balance your activity with adequate relaxation and sleep so that your tissues can rejuvenate properly.
Vata Season Yoga
Incorporating a sense of warmth, grounding, stability, and focus on your yoga practice has a profoundly calming effect on Vata and can work wonders during the Vata season. Your breath should be deep and fluid. If you practice pranayama (yogic breathing exercises), alternate nostril breathing is very balancing this time of year. In your asana practice, favor Vata-pacifying yoga. Warm-up slowly and include some joint rotations. Move with intention and fluidity—grounding the hands and the feet on the mat whenever possible—and avoid jumping between postures. Gentle flows like a relaxed sun salutation are perfect for Vata. You can also favor standing and balancing poses such as mountain, warrior I, warrior II, and tree pose to increase stability and strength. Connect with the earth beneath you in poses such as thunderbolt, cat-cow, cobra, and child’s pose, and quiet the mind with forwarding bends such as intense westward stretch. Gentle inversions and restorative poses such as legs up the wall are also very good for Vata. Close your practice with a long corpse pose, covering yourself with a blanket so that you don’t get chilled.
Herbal Support for Vata Season
Taking Chyavanprash in the morning can help to reinforce immunity, strength, and energy during the autumn season. Ashwagandha is stabilizing to the mind and nervous system, and can promote sound sleep, strong digestion, proper elimination, and appropriate strength; it is available as a powder, tablet, and liquid extract. Similarly, herbal teas made from ginger, licorice, or a combination of cumin, coriander, and fennel, can help to promote proper digestion and warmth. Another grounding, vitalizing herbs and formulas include Dashamula, Haritaki (also available in tablets), Triphala (also available in tablets), and Vidari. The following herbal tablets are also generally quite supportive during the Vata season: Healthy Vata, Joint Support, Mental Clarity, Stress Ease, Tranquil Mind, and Vata Digest.
More Specific Support for Your System
The following links to dosha-specific recommendations are intended to assist you in offering more personalized support to your particular constitution. If you don’t know yours, consider taking our simple Prakriti quiz, and then choose the appropriate link below to further customize your seasonal routine.
Remember, a seasonal routine is an investment in your own health and vitality. And while the specifics may vary from one person to the next, we all stand to benefit from aligning ourselves with the rhythms of nature throughout the year. This fall, embrace the unique gifts of autumn and—with the help of an appropriate seasonal routine—enjoy it from a place of stability, humility, and gratitude.
Ayurveda is an ancient science based on elemental principles that pertain to life on earth. Ayurveda recognizes the elements of ether, air, fire, water, and earth as the building blocks of the natural world. According to Ayurveda, these five elements pair-up in three combinations to form the primary forces of nature called doshas. Ether and air from Vata dosha. Fire and water make up pitta dosha. Water and earth create Kapha dosha.
Under the influence of Vata’s ether and air contributions, you can feel light, carefree, and creative or spacy, scattered, and unstable. The etheric nature of Vata creates a sense of space, in which you may feel free or lost. The airy aspect of Vata can inspire productivity or promote anxiety. Ayurveda teaches that like increases like. If you are dominantly Vata by nature or are consistently influenced by Vata, you are more likely to experience the negative effects of excess Vata during the Vata season.
As the external environment changes during the Vata season, your internal environment can experience the same type of changes; dry leaves, dry skin; crackly leaves, crackly joints; shorter days, shorter attention span; colder days, colder extremities, windy days, windy bowels. The qualities of Vata dosha are found in the disorders that are common at this time of year. By observing the processes of Mother Nature, you can better understand the processes of your body, mind, and spirit.
Applying the Ayurvedic principle that opposite actions create balance, you can maintain balance during the Vata season by emphasizing lifestyle and food choices that are grounding, stabilizing, warming, moisturizing, and softening. You can stay calm and connected in this whirlwind season with a consistent practice that includes nourishing and protective measures. Ayurveda promotes simple and regular routines as having a deeper effect on balancing Vata than an ‘as needed’ approach.
Offered here are Ayurvedic recommendations for enjoying the Vata season with stability and serenity